 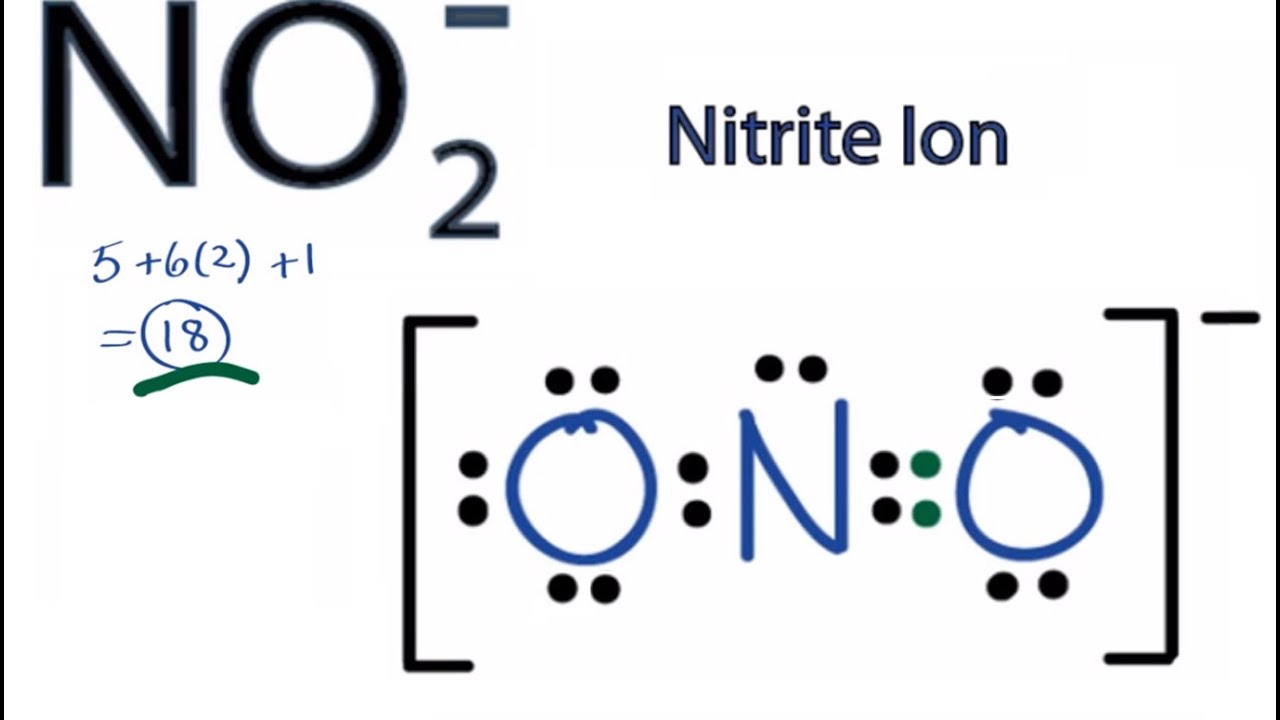
VESPR theory is responsible for deciding the presence of bond pairs and lone pairs in compounds and its effect on the shape. The shape of compound depends on few factors such as the present of bond pairs and lone Pairs in the compound impacts on the sculpture of the compounds. Therefore, NO2 has two consecutive resonating structures. The π bond opens up and generates negative charge on another oxygen atom and vice versa. There is one extra electron present on one oxygen atom that creates bond with the centre atom in NO2 that is Nitrogen. After drawing the complete Lewis structure of NO2, it has been found that one oxygen atom holds one extra valance electron, which cannot get scope to undergo bonding. The number of valance electrons decides formation of bond pairs and existence of lone pairs in the compounds. The most electronegative atom is placed in the centre that is the Nitrogen in NO2 is considered as centre atom of this compound. Moreover, several factorial characteristics can be represent by the following the steps of drawing. Then placing g the suitable atom in the centre of the compound one can draw the basic Sketch of the NO2 Lewis structure. In the first place calculating the number of Valence electron is the most significant step to follow. Those steps give specific structure with proper geometric knowledge. This article will represent those facts such as:ĭrawing Lewis structure of Nitrogen dioxide is simple and followed by the fundamental steps of Lewis structure formation. Many facts about its internal structure can be obtained from the drawing of Lewis did structure. NO2 Lewis structure shows several chemical properties of the compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
